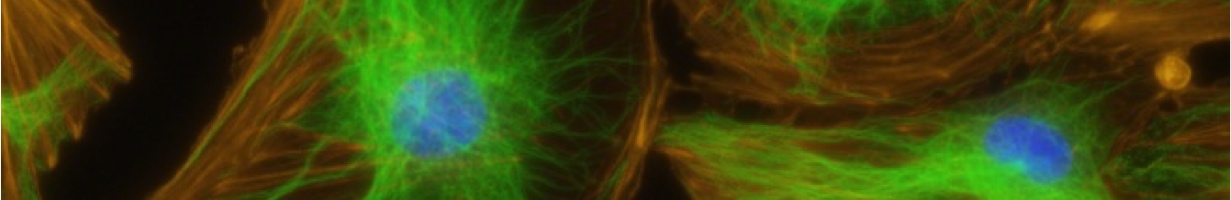
Introduction to the history of QCM

Invitrometrix designs and manufactures a multi-well Quartz Crystal Microbalance (QCM), a sensitive surface sensing technology.
Our technology is designed to advance Life science & Drug discovery research, Chemistry and Biomaterial research in a multi-well based format.
Why Use QCM?
Established Precision Device

QCM has been used as a precision device measurement in the ng range for the applications
Once Tuned:
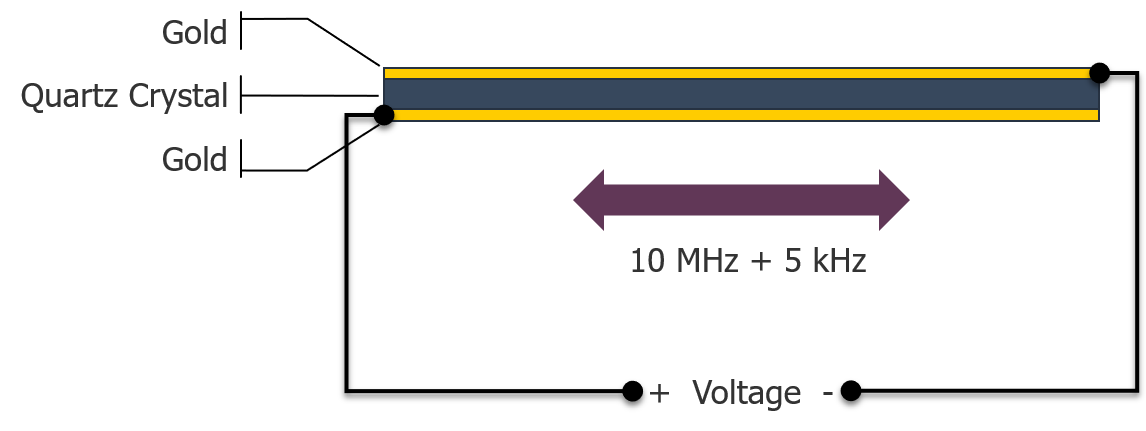
Following the development of the Sauerbrey equation in 1959, oscillation changes in frequency could be correlated to change in mass on the microbalance’s surface and was subsequently used to measure gas or elements in a vacuum. A QCM is made up of some basic components: an AT cut piezoelectric quartz crystal and metal electrodes. When a voltage is applied across the crystal this causes the crystal to oscillate at a fixed rate. Invitrometrix uses 10 MHz crystals as its sensing element.
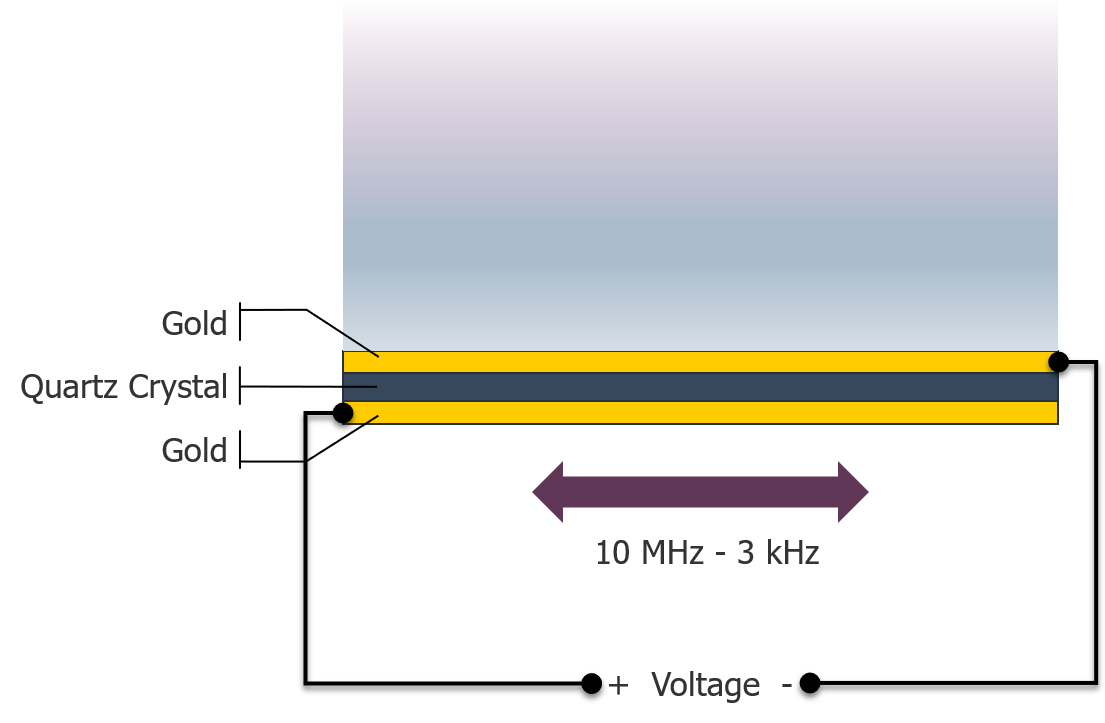
To enable research to occur in a liquid environment, the Kanazawa-Gordon equation is applied. This enables research in chemistry and biochemistry applications. This enabled the measurement of enzymatic reactions, antibody binding. More recently the QCM has been used to measure changes in anchorage dependent whole cells. Previous research has shown that a QCM device is able to measure anti-body based immunoassay by direct or sandwich methods, detection of nucleic acids, glucose detection via hexokinase binding, real time measurement of cell metabolism and division rates, and immunological detection of microbes.
Invitrometrix Corporation has developed the InVitro-Q, a modified QCM multi-well device that can monitor real time changes occurring within living cells while maintaining physiologically appropriate temperature, humidity, and CO2. The Discovery-Q is our first commercial system with a 4-well miniplate format.